What is Spermidine?
 Spermidine is a naturally occurring polyamine, having two or more primary amino groups, and is widely encountered in ribosomes and living tissues and is critical for cell survival and function. In the body spermidine is created from its precursor putrescine. Spermidine is the precursor to further polyamines, such as spermine, which is also critical to cellular function.
Spermidine is a naturally occurring polyamine, having two or more primary amino groups, and is widely encountered in ribosomes and living tissues and is critical for cell survival and function. In the body spermidine is created from its precursor putrescine. Spermidine is the precursor to further polyamines, such as spermine, which is also critical to cellular function.
Dutch scientist Anton van Leeuwenhoek, one of the early pioneers of Microscopy, first identified spermine in human semen back in 1678. Likewise, spermidine was first discovered in human sperm.
Spermidine is one of a number of natural substances that have been described as geroptrotectors, substances that may slow down the aging process.
Sources of spermidine
Dietary sources of spermidine are grapefruit, mature cheese, mushrooms, soy products, legumes, corn, and whole grain, chickpeas, peas, green peppers, broccoli, oranges, green tea and rice bran. However there are currently no natural spermidine extracts on the market.
Dietary supplement manufacturers have developed synthetic spermidine to emulate its natural counterpart. Synthetic spermidine replicates the molecular structure of natural spermidine and is mechanistically identical to that found in nature. Thanks to modern manufacturing techniques, synthetic spermidine is one hundred percent biologically identical to the chemical structure of the spermidine found in nature. Furthermore, synthetic spermidine provides a product with a potency that cannot be obtained through dietary sources.
Spermidine is one of the more interesting polyamines and research suggests that spermidine and its derivatives interact with various cellular processes to promote health and may even delay aging.
Biochemistry of putrescine
 Before we talk more about spermidine we should understand where it comes from and the events leading up to its creation. The story starts with putrescine and a chain of events that lead to the creation of spermidine.
Before we talk more about spermidine we should understand where it comes from and the events leading up to its creation. The story starts with putrescine and a chain of events that lead to the creation of spermidine.
Putrescine is the precursor of spermidine and creates it as part of a process during which putrescine attacks decarboxylated S-adenosylmethionine. The interaction of living cells and ornithine decarboxylase also results in the synthesis of small amounts of Putrescine.
Putrescine synthesis occurs via two unique pathways, but the process begins with arginine in both instances.
Arginine is converted into agmatine in one pathway with the assistance of an enzyme called arginine decarboxylase (ADP). Agmatine is then converted into N-carbamoyl putrescine by agmatine imino hydroxylase. The series of biological processes comes full circle when N-carbamoyl putrescine is transformed into putrescine. The second pathway promotes the conversion of arginine into ornithine. Ornithine is then transformed into putrescine by a compound called ornithine decarboxylase. Putrescine is a simple polyamine and it appears that polyamines function in a similar way to growth factors during cell division.
One form of macromolecular degradation is a cellular process known as autophagy, which plays a significant role in the elimination of mitochondria. Polyamines such as spermidine and putrescine are known to promote autophagy. Researchers have made enormous progress toward understanding the nature and mechanisms associated with mitochondrial degradation in recent years. It appears that the degradation of mitochondria through the mechanism of autophagy is more tightly regulated than was previously believed (1).
Biochemistry of spermidine
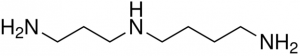 The chemical formula for spermidine is C7H19N3 and it is one of the polyamines, organic compounds having two or more primary amino groups. Polyamines are polycations that will interact readily with negatively charged molecules, including DNA, RNA and lipids. The ability of polyamines to bind so many different types of molecules gives them great utility and allows them to be be involved in various processes including, cell growth, DNA stability, cell proliferation and apoptosis (2).
The chemical formula for spermidine is C7H19N3 and it is one of the polyamines, organic compounds having two or more primary amino groups. Polyamines are polycations that will interact readily with negatively charged molecules, including DNA, RNA and lipids. The ability of polyamines to bind so many different types of molecules gives them great utility and allows them to be be involved in various processes including, cell growth, DNA stability, cell proliferation and apoptosis (2).
Spermidine plays a key role in the synchronization of various biological processes, such as levels of intracellular pH and the maintenance of membrane potential. Spermidine has also been shown to regulate a number of important biological processes, including aspartate receptors, nitric oxide synthase, cGMP/PKG pathway activation and cerebral cortex synaptosome activity.
Spermidine is the precursor to a number of other polyamines, such as spermine and its structural isomer thermospermine. Spermidine is of significant interest to scientists researching aging because it is a key morphogenetic determinant for the life span of cells and living tissues (3). Spermidine has been shown to have pro-longevity effects in mammals due to various mechanisms of action.
Spermidine may slow down the aging process
Recently spermidine has been in the spotlight due to its potential as a geroprotector that may potentially delay the aging process. The ability of spermidine to enhance autophagy is thought to be the main geroprotective mechanism that slows down the aging process at the cellular level (4).
Autophagy is a recycling process for cells and allows the destruction and recycling of unnecessary and damaged molecules or even entire organelles. A defective autophagy mechanism has been associated with a host of age related diseases. Spermidine has been shown to induce autophagy in mouse liver cells, worms, yeast and flies (5). On the other hand, a lack of spermidine and autophagy is highly correlated with reduced life spans and the detrimental effects of stress and acute inflammation.
Despite autophagy being the primary mechanism of spermidine, evidence has also been found for other mechanisms, including anti-inflammatory (6-7) properties, lipid metabolism, regulation of cell growth and proliferation (8-9), and apoptosis (10) to play a role in its geroprotective properties.
Excessive inflammation levels can lead to cell damage that can promote age-related diseases and as we get older chronic inflammation levels rise as part of that process. The more inflammation at the cellular level your body has, the faster your cells are being destroyed which contributes to a more haggard and aged appearance. Researchers found that polyamines, in particular spermidine, can reduce this inflammation thus slowing down one driver of the aging process. Since spermidine targets the source of inflammation, it could protect your cells from the damage inflammation causes during aging.
Supplementation of spermidine has been shown to increase lifespan in a number of animal studies (11- 12) as does a diet rich in polyamines (13). There is also evidence to suggest that spermidine improves resistance to stress and that declining levels of spermidine with age drive the aging process leading to onset of age-related diseases (14-15).
Lipid metabolism is also known to be a regulator of health and lifespan and a dysfunction in the lipid metabolism can have serious consequences on health, aging and lifespan. The role of spermidine in adipogenesis and its ability to modify lipid profiles makes lipid regulation another likely reason why spermidine affects lifespan.
At the cellular level, spermidine facilitates the differentiation of preadipocytes into mature adipocytes as part of the adipogenesis process. A study showed that treatment with α-difluoromethylornithine (DFMO) an inhibitor of polyamine synthesis was able to completely halt adipogenesis and thus disrupt lipid metabolism (16-17). Treatment with spermidine caused the resumption of differentiation in the presence of DFMO as well as restoring expression of transcription factors key in preadipocytes differentiation as well as late adipocyte markers.
Another mechanism that lends itself to the geroprotective potential of spermidine is the cellular growth spurred by it. When normal cell growth is not being inhibited, tissue is able to regenerate faster, leading to smoother and more youthful looking skin. Research shows that spermidine levels decline as you age causing cell growth to decline. With additional infusion of spermidine, cellular growth can proliferate, potentially leading to a slowing of the aging process.
Taken together effective autophagy, reduction of inflammation, lower stress levels in the cell, improved cell growth and improved lipid metabolism could all potentially play a role in slowing down the aging processes as well as improving health generally.
How to take spermidine
In order to get the best results from using spermidine we recommend using a quality supplement that has been manufactured in accordance with rigorous quality controls and laboratory analysis.
Reputable dietary supplement manufacturers will clearly describe the method they use to manufacture their spermidine supplements. Spermidine supplements may be offered in a pure form that contains no additives, or in the form of a compound that includes other active ingredients that work in synergy.
The use of of spermidine as an accompaniment to food should not cause any health related side effects if the recommended dose and treatment instructions are strictly observed. As with all supplements they are not intended to replace a healthy and balanced diet.
As applies to taking any supplements, certain individuals, especially young children, pregnant and breastfeeding women and patients using medication are advised to consult their physician prior to taking spermidine. If you experience symptoms of weakness, abdominal pain, dizziness, skin irritation or other side effects while taking spermidine, stop taking all nutritional supplements and consult your physician.
References
(1) Goldman, S. J., Taylor, R., Zhang, Y., & Jin, S. (2010). Autophagy and the degradation of mitochondria. Mitochondrion, 10(4), 309-315.
(2) Minois, N., Carmona-Gutierrez, D., & Madeo, F. (2011). Polyamines in aging and disease. Aging (Albany NY), 3(8), 716-732.
(3) Deeb, F., van der Weele, C. M., & Wolniak, S. M. (2010). Spermidine is a morphogenetic determinant for cell fate specification in the male gametophyte of the water fern Marsilea vestita. The Plant Cell, 22(11), 3678-3691.
(4) Eisenberg, T., Knauer, H., Schauer, A., Büttner, S., Ruckenstuhl, C., Carmona-Gutierrez, D., … & Fussi, H. (2009). Induction of autophagy by spermidine promotes longevity. Nature cell biology, 11(11), 1305-1314.
(5) Eisenberg, T., Knauer, H., Schauer, A., Büttner, S., Ruckenstuhl, C., Carmona-Gutierrez, D., … & Fussi, H. (2009). Induction of autophagy by spermidine promotes longevity. Nature cell biology, 11(11), 1305-1314.
(6) Bjelaković, G., Stojanović, I., Stoimenov, T. J., Pavlović, D., Kocić, G., Rossi, S., … & Bjelakovic, L. J. (2010). Metabolic correlations of glucocorticoids and polyamines in inflammation and apoptosis. Amino acids, 39(1), 29-43.
(7) Choi, Y. H., & Park, H. Y. (2012). Anti-inflammatory effects of spermidine in lipopolysaccharide-stimulated BV2 microglial cells. Journal of biomedical science, 19(1), 31.
(8) Landau, G., Ran, A., Bercovich, Z., Feldmesser, E., Horn-Saban, S., Korkotian, E., … & Kahana, C. (2012). Expression profiling and biochemical analysis suggest stress response as a potential mechanism inhibiting proliferation of polyamine-depleted cells. Journal of Biological Chemistry, 287(43), 35825-35837.
(9) Landau, G., Bercovich, Z., Park, M. H., & Kahana, C. (2010). The role of polyamines in supporting growth of mammalian cells is mediated through their requirement for translation initiation and elongation. Journal of Biological Chemistry, 285(17), 12474-12481.
(10) Minois, N., Carmona-Gutierrez, D., & Madeo, F. (2011). Polyamines in aging and disease. Aging (Albany NY), 3(8), 716-732.
(11) Yue, F., Li, W., Zou, J., Jiang, X., Xu, G., Huang, H., & Liu, L. (2017). Spermidine prolongs lifespan and prevents liver fibrosis and hepatocellular carcinoma by activating MAP1S-mediated autophagy. Cancer Research, canres-3462.
(12) Eisenberg, T., Abdellatif, M., Schroeder, S., Primessnig, U., Stekovic, S., Pendl, T., … & Tong, M. (2016). Cardioprotection and lifespan extension by the natural polyamine spermidine. Nature medicine.
(13) Soda, K., Dobashi, Y., Kano, Y., Tsujinaka, S., & Konishi, F. (2009). Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Experimental gerontology, 44(11), 727-732.
(14) Minois, N., Carmona-Gutierrez, D., Bauer, M. A., Rockenfeller, P., Eisenberg, T., Brandhorst, S., … & Madeo, F. (2012). Spermidine promotes stress resistance in Drosophila melanogaster through autophagy-dependent and-independent pathways. Cell death & disease, 3(10), e401.
(15) Landau, G., Ran, A., Bercovich, Z., Feldmesser, E., Horn-Saban, S., Korkotian, E., … & Kahana, C. (2012). Expression profiling and biochemical analysis suggest stress response as a potential mechanism inhibiting proliferation of polyamine-depleted cells. Journal of Biological Chemistry, 287(43), 35825-35837.
(16) Vuohelainen, S., Pirinen, E., Cerrada‐Gimenez, M., Keinänen, T. A., Uimari, A., Pietilä, M., … & Alhonen, L. (2010). Spermidine is indispensable in differentiation of 3T3‐L1 fibroblasts to adipocytes. Journal of cellular and molecular medicine, 14(6b), 1683-1692.
(17) Hyvönen, M. T., Koponen, T., Weisell, J., Pietilä, M., Khomutov, A. R., Vepsäläinen, J., … & Keinänen, T. A. (2013). Spermidine promotes adipogenesis of 3T3-L1 cells by preventing interaction of ANP32 with HuR and PP2A. Biochemical Journal, 453(3), 467-474.
